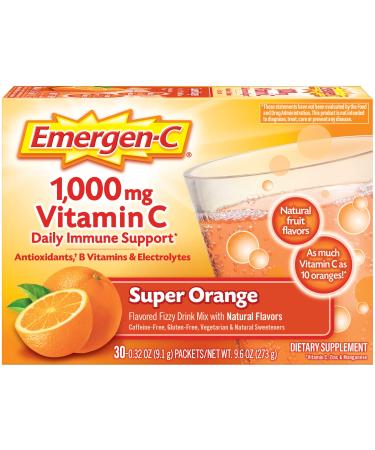
*Based on studies: Djuist AMS Scar Reduction Dressing Clinical Evaluation 2004
Bieley et al. Clinical Paper_Effects of a water-resistant, non-silicone-based occlusive dressing on keloids. J Am Acad Dermatol 1996;35:114-6, Jul 1996
Bisbal J. Efficacy of a polyurethane self-adhesive dressing in preventing hypertrophic scars. Plastic circl. iberolatinoam.-Vol. 37 - No. 4. October - November - December 2011 / Pag. 341 - 347
Precautions and contraindications: redness may occur. If redness, pain and/or irritation occurs, discontinue use of Trofolastin Scar Reducer and consult a doctor or healthcare professional. Do not use on 3rd degree burns. Do not use on open wounds, infected wounds, scabs, over stitches or staples. Do not use if the container is open or damaged. Not suitable for patients with dermatological alterations where skin integrity in the area of application is affected. Do not use in case of allergies to acrylics or polyurethanes. Do not use in newborns. Do not use with oxidising solutions such as hypochlorite or hydrogen peroxide as they may break down the polyurethane of the dressing. Single use: once opened, the product should not be reused due to the risk of contamination and infection. If long-term repeated use is required, the clinical assessment of a doctor/healthcare professional. For external use only.
Trofolastin Elasticity scar reducer complies with current regulations for health products. C 2019 Group of companies GSK or licenser. The trademarks are the property of or are licensed to the GSK Group of companies. CPS 3254-PS-CM / CHESP/CHCARR/0004/19








![Stewart Freeze Dried Dog Treats Made in USA [Single Ingredient Puppy and Dog Training Treats - Grain Free Natural Dog Treats] Resealable Tub to Preserve Freshness - Buy Online on GoSupps.com](https://www.gosupps.com/media/catalog/product/cache/25/small_image/375x450/9df78eab33525d08d6e5fb8d27136e95/6/1/61gwbbixarl._ac_sl1500_.jpg)